Medical Coolers That Go the Distance
Transport supplies worry-free with lab-qualified vaccine and medical travel coolers that provide safe storage and maintain stable temperatures for up to three days.
Keep Your Cool
Get a lab-qualified medical cooler (also called vaccine carriers) to transport supplies and support your emergency preparedness plans.
Up to 3 Days of Cold-Chain Protection
Count on days of stable temperatures within the critical 2-8°C range (when the cooler is kept closed).
Hours of Stability for Offsite Use
Reduce supply loss with eight hours of temperature control when the cooler is opened and closed.
Secure, Ergonomic Design
Travel worry-free with carrier features including a lightweight cart, durable carrier material, and easy access options.
Versatile and Effective
Provide reliable medical-grade storage and transport for blood, medicine, and more with no ice packs or electricity required.
TempArmour® medical coolers use phase-change material (PCM) and vacuum-insulated panel (VIP) technology to improve emergency preparedness and deliver better cold-chain protection during transport and for off-site care.
TempArmour Medical Cooler (Model VCT-4)
The VCT-4 Carrier includes panels to maintain vaccines within the refrigerated range (2-8°C) for days without a backup power source.
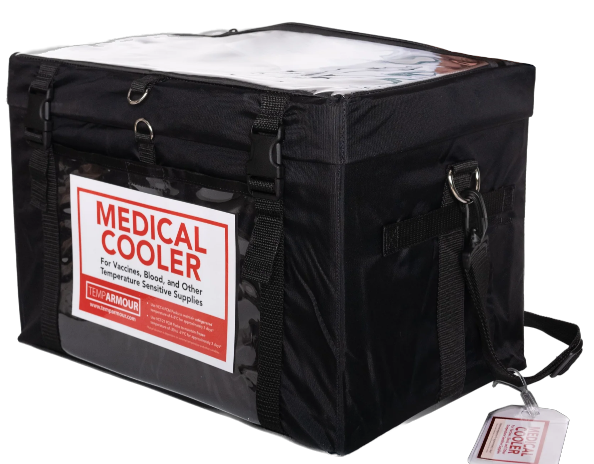
Temperature Control When and Where You Need It

Steady on the Go
Know your timelines and requirements to transport temperature-sensitive supplies in the event of a storm, outage, or emergency.
TempArmour Medical Cooler (Model VCT-21)
The VCT-21 Carrier includes the same convenient, reliable design features as the VCT-4, with panels that maintain vaccines within the frozen range (-50°C to -15°C) for daily and emergency use.


Cold-Chain Storage That’s Always on Call
Maintain stable temperatures for up to six days during storms and outages with medical-grade refrigerators and freezers for doctor’s offices, vaccine clinics, and pharmacies.
Better Protection for a Better Experience
“I received the carrier and I love it already! It is compact and I really like how it comes with the strap and wagon for easy transportation.”
“This is a game-changer for our vaccine clinic administrations.”
When Every Dose Counts
Transport and provide temperature-sensitive supplies where they’re needed – no ice packs required.

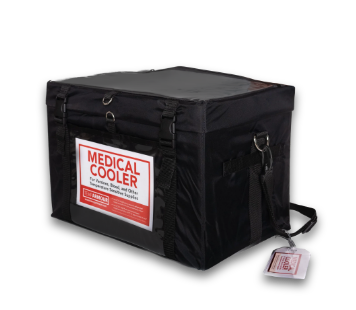
.png?width=774&height=772&name=CarrierPanelsClosed_30a42545-8123-4e5d-b095-05792913a471_1024x1024@2x%20(1).png)



